Meeting Cold Chain Challenges of Patient-Specific ATMPs
Leading Minds Network Panel Discussion:
The following summary is from a panel discussion hosted by the ELPRO Leading Minds Network as part of its Temperature Control Tuesdays™ programming in partnership with Cold Chain Platform and PCI Pharma Services. This session featured Hedley Rees of PharmaFlow Ltd., Jackie (Heneghan) Peck of Pharmacy Consulting, and Gavin Morgan of PCI Pharma Services.
The rapidly expanding biotech sector of cell and gene therapies is not merely creating new medicine; it is literally creating a new approach to healthcare across the globe. A 2021 highlight report published by the American Society of Gene and Cell Therapy in connection with Pharma Intelligence estimated 3,400 cell and gene and RNA therapies were in the pipeline globally. A forecast published by Cell and Gene Therapy Catapult indicated that by the year 2024, the demand for new jobs in the sector will double today’s workforce. Additionally, the projected skills demand for bioprocessing jobs in 2026 is 10,000, up 151% from where it stands today. Advanced therapy medicinal products (ATMP) includes gene and cell therapy as well as tissue engineered products.
ATMPs include the following biologics:
- Allogeneic products are not specific to the patient. They can help any of the patient population that have a particular condition that needs some modification.
- Autologous products are specific to the patient. The patient is the source of the product, and the same patient uses the product.
The panel discussion focused on the complex supply chain for autologous products. Jackie (Heneghan) Peck of Pharmacy Consulting expects growth specifically for autologous products because there are fewer side effects when the patient’s cells are used rather than using the cells of a donor.
Hedley Rees, chief of Pharmaflow Ltd., described the process of an autologous clinical trial. He explained that the doctor and patient will decide if a specific therapy is required at an appointment at the hospital. Typically, the CAR T-cell therapies are advanced in the blood cancers. At the hospital site, patient cells are taken, and the process of apheresis is carried out where the cells are cryogenically frozen. The cells are then picked up at the site, and the full pedigree of traceability of the cells begins. If the wrong cells go back into the patient, it means certain death. The patient is both the source and the destination of the supply chain.
Rees believes that the novel, patient-specific ATMPs (autologous) require one of the most extreme supply chain challenges that the world has ever known. ATMPs frequently move to the next steps of the supply chain while testing is ongoing. Therefore, it’s important to ensure that all details are considered prior to re-administering to that patient. From a logistics standpoint, there is a lot of work done up front.
Managing the supply chain logistics is difficult because human cells are extremely sensitive to time and environment. According to Gavin Morgan of PCI Pharma Services, there is a very small window of opportunity in this process. To make things more complex, there are also many key stakeholders in the supply chain, including physicians, couriers, and third party logistics (3PL) providers. Each additional handler increases the risks to a successful delivery. The graphic below illustrates the life cycle of CAR T-cell infusion.
Chart courtesy of Hedley Rees, PharmaFlow Ltd.
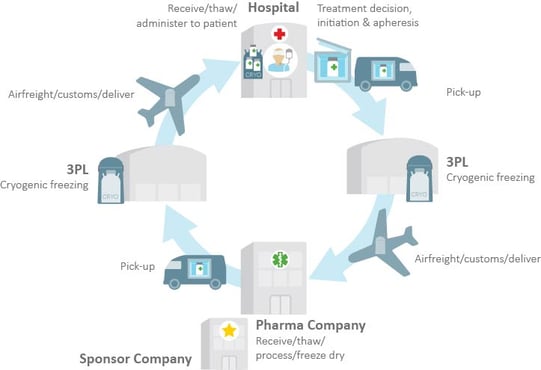
N.B. 3PL = Third Party Logistics Provider
Letting academic medical centers make CAR-T drugs (see useful links): “It talks the company two to six weeks to engineer the cells, increase their number, perfom quality and safety tests, and ship them back to the hospital to be reinfused into the patient”
Throughout the discussion, the panelists concentrated on three major challenges and how to overcome them.
Challenge 1: Finding the Right Clinical Trial Site
Morgan noted that many sponsors conduct feasibility studies to determine which sites they're going to utilize. The sites need to be able to handle just-in-time (JIT) deliveries. It should be feasible to collect the sample and get it to where it needs to go within the required timeframe. According to Morgan, the Asia-Pacific (APAC) region commonly ships samples from one country to another, where there are so many countries in close proximity to each other. Entry into some countries are more time-consuming than others. “Make certain you have what is required and you can receive authorization in time for the samples to arrive at its destination safely,” he said Morgan also recommended testing ports of entry to find how to bring transit time down to a minimum.
Challenge 2: Finding the Right Manufacturer
Sponsors should look to manufacture as close to the patient as possible. Morgan said it would be great if hospitals could do it literally on campus or close, but that is not going to happen overnight. At this time, these kinds of studies aren't going to happen in a remote region, where manufacturing locations are not close in proximity and/or there is no logistics framework in place. He also expressed the need for more manufacturing sites closer to the source, spread globally to reduce the need to collect in the APAC region.
“Because biologics are more difficult in terms of manufacturing small molecule products, industrial chemistry, we don't have much choice as to what plant you use. In a sense, there needs to be a whole sort of re-engineering mindset around this,” said Rees.
“Even if the study is performed in a developed location, you can’t expect just any manufacturer to be proficient at genetic modification and handling biologics, living things, cells, etc. The processes of determining stability, doing freeze thaw studies, and the whole release process for these products is a challenge,” Rees noted. He recommended researching which companies are most efficient. The company must be able to implement good distribution practice and good manufacturing practice thoroughly and quickly.
Challenge 3: Delivery Interruptions
Advance planning and coordination requires backwards planning. Once you’ve identified the locations in which products will be shipped, you need to research which shipping systems are able to make the deliveries within the predetermined timeline. Peck recommended companies to do a test run on the prepared route in advance. In doing so, one can confront probable challenges before the actual product is compromised.
Each panel member reiterated the necessity that all stakeholders and their backups understand their roles, responsibilities, and the timelines required for a successful delivery. Legibility is a problem within supply chains. If the process requires a legible documented signature, it will be easy to trace the product at all times. Additionally, the temperature monitoring loggers that trace geographic location should accompany the product from the starting point to the point of delivery (POD).
Deviations
“Things do go wrong with logistics and hospitals, especially in a pandemic,” Peck said. With ATMPs, there isn’t enough time to do detailed investigations and approval for every excursion like there is for other products. In advance, responsible parties must have the specified temperature ranges to which medicines can be safely administered.
Documentation
Many different documents and licenses are required if you are exporting blood samples for further manufacturing, versus dispatching blood samples for testing.
Morgan warned, “What you don’t want is a sample like this to land in country and just sit at the port of entry.”
The products must pass quality checks. Morgan added that there are times when customs documentation should state that someone is bringing that shipping system back for reuse. “It needs to be listed on the pro forma invoice with a value associated to it. These are all responsibilities of your CMO or the 3PLs. In some countries, there are workarounds. In other countries, it is only feasible to manufacture locally,” he said.
For example, Morgan noted that China has strict rules regarding the export of any human tissue or blood samples. The export licenses needed to get material like this out of China can take two or three months to obtain. Singapore, however, is less challenging.
If bringing material into the US, these types of products are governed through the FDA and customs in the same way as any kind of clinical trial supply with GMP and GDP. Morgan noted that, if 3PLs are a known consignor, they may be able to avoid having samples x-rayed and speed up the exporting process.
Delivery Complexity
The products should be delivered specifically to the person that's expecting it, not simply dropped off as goods at a dock. The collection and receipt should have two or three names of personnel (backups if the others are unavailable) that can sign for the material, and their sample signatures to ensure authenticity.
Rees noted that different hospitals have different rules. Typically, the pharmacy is the gatekeeper into the hospital, and there are specific rules about how products are released into the hospital. Your vehicle may require special access to the facility and/or where it should be parked. The collection and the receipt should state the delivery location, but the driver can easily be confused. Even if they are familiar with the hospital, there may be a warehouse outside of the facility where the delivery should be made instead. Peck added that drivers must be informed, trained, and completely briefed as to what they need to do rather than just relying on a label or a drop sheet.
During the pandemic, Morgan mentioned that couriers arrived at the hospital only to be restricted access because of COVID-19. Couriers may even need pre-authorization or specific instructions to gain access to the right department, and the department should be expecting them.
There are other regulations to consider as well. The EU General Data Protection Regulation (GDPR) should be able to determine what you can and cannot share or how it's coded or bar coded.
Retrieval Complications
Clinics and hospitals are familiar with blood samples that are refrigerated. They may even be used to the 15 °C to 25 °C, even dry ice shipments, but they may not be used to receiving LN2 shippers. Cell and gene therapies are often shipped in liquid nitrogen. Sites should be prepared to safely handle the shipper and product. Otherwise, you need to assign a trained courier to handle the shipment.
Peck finds it exceptionally difficult to ensure employees are thoroughly trained on safety throughout the supply chain and then to maintain the competent workers amid the busy environment.
COVID-19 Effects on Supply Chains
When products are in high demand, it demonstrates the importance of a functional supply chain. The capacity of the supply chain is limited to the bottlenecks. Rees explained how COVID-19 created bottlenecks in unexpected places and forced people to manage them effectively. The huge vaccine rollout has actually improved standards in many sites because they had deadlines and expectations to meet.
Peck added that companies had to create disaster plans in case of employee unavailability, extra training plans for health and safety to make sure they could still maintain compliance. More people are now able to handle a variety of containers and packaging materials.
Morgan described how COVID-19 forced companies to implement new backup plans. Because of passenger shortages, commercial flights were frequently canceled at the last minute. Commercial flights make money on passengers, not cargo, so couriers needed an alternative way to make deliveries. Some couriers chartered their own flights because cargo flights generally do not meet the required timelines for ATMPs.
Final Thoughts
Process expense comes from a shortage of patients, inconvenient manufacturing locations, and the complex supply chain. Stakeholders need to come together and optimize the process in order to make products available and affordable.







Leave a Comment